|
All isotopes of an element have the same chemical properties. are atoms of the same element with different numbers of neutrons and therefore different masses. And their natural abundances are 75.78 and 24.22, respectively. Calculating Average Atomic Mass Topic Pennies can be used as a model for atomic isotopes and for calculating average atomic mass. Their atomic masses are 34.96885 amu and 36.96590 amu. What is the average atomic mass of chlorine The two stable isotopes of chlorine are Cl and Cl. To use this resource Click Here.Ī useful resource, written by Jef Rozenski, calculates all possible combinations of H, C, N & O that give a specific nominal mass. The resultant value is the average atomic mass of the element. Of course, compounds of chlorine and bromine have very large isotope abundances.Ī more elegant and complete isotope pattern calculator has been created by Junhau Yan. 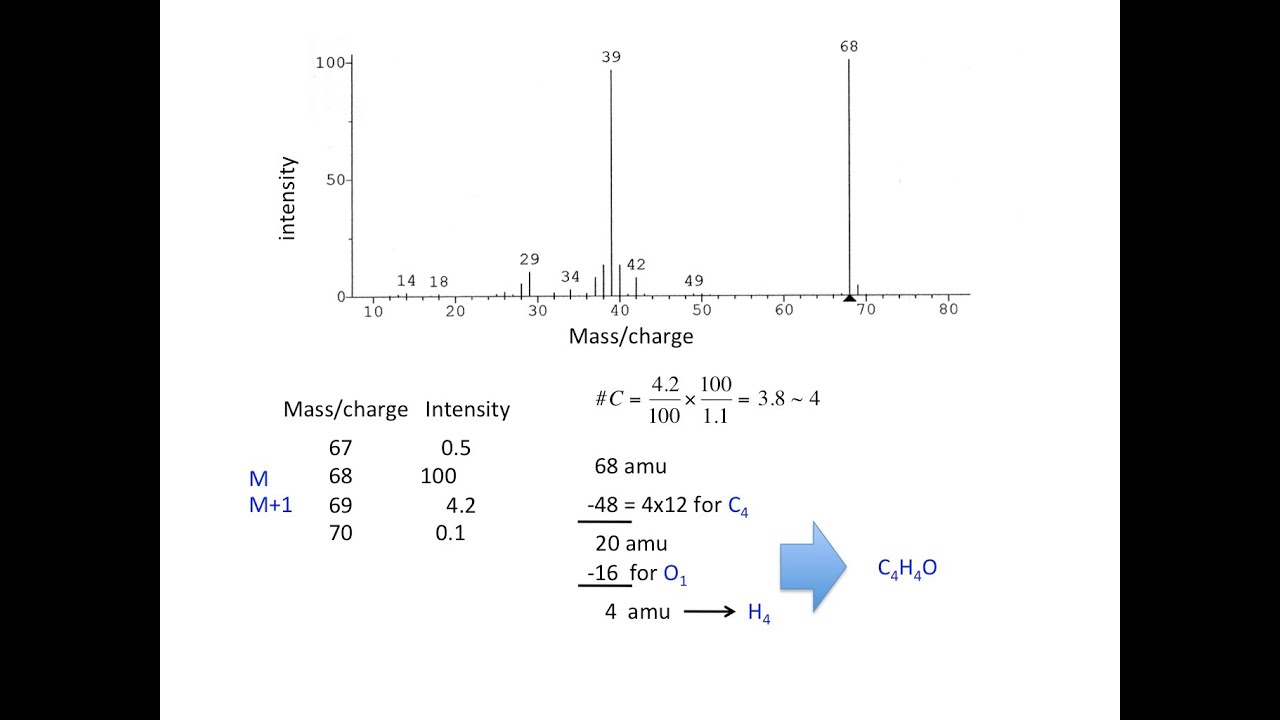
The numbers displayed in the M+1 and M+2 boxes are relative to M being set at 100%. Simply enter an appropriate subscript number to the right of each symbol, leaving those elements not present blank, and press the " Calculate" button. 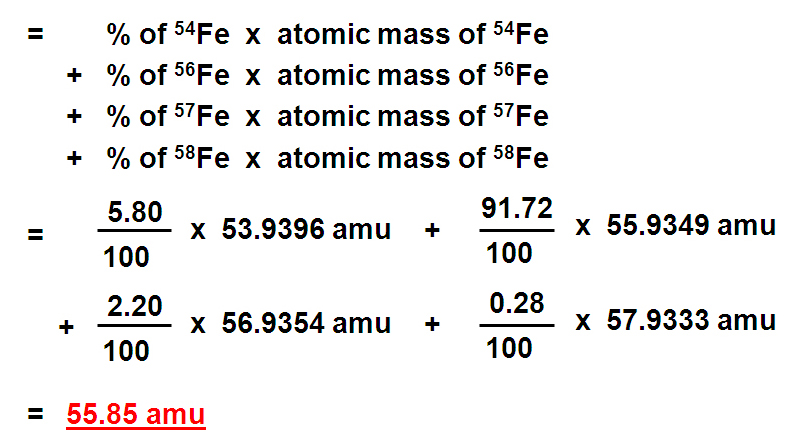
The calculator on the left may be used to calculate the isotope contributions to ion abundances 1 and 2 amu greater than the molecular ion (M). Use the table below to find the atomic weight of each atom (element), or refer to a Periodic. The average Atomic Mass of various elements are determined by multiplying the Atomic Mass of each isotope by its fractional abundance and adding the value. For compounds of chlorine and bromine, increments of 1.997 and 1.998 respectively must be added for each halogen to arrive at the higher mass isotope values. Let us calculate the molecular weight of some common compounds. Only the mass of the most abundant isotope, relative to C (12.0000), is used for these calculations. The mass calculator on the right may be used to calculate the exact mass of a molecule based on its elemental composition. 
This table is adapted from Introduction to Mass Spectrometry, by J.T. M i molar mass i (g/mol) The number average molar mass is heavily influenced by small molar masses that are likely to be in larger numbers. The factor is multiplied by the number of atoms (n) of the designated element to calculate the intensity contribution from higher mass isotopes. * X represents the relative intensity of the lowest mass ionin an isotopic ion cluster. Masses Exact Masses & Isotope Abundance Ratios Element
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
